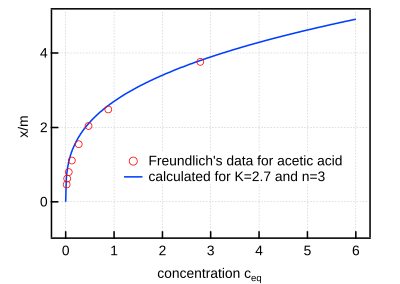
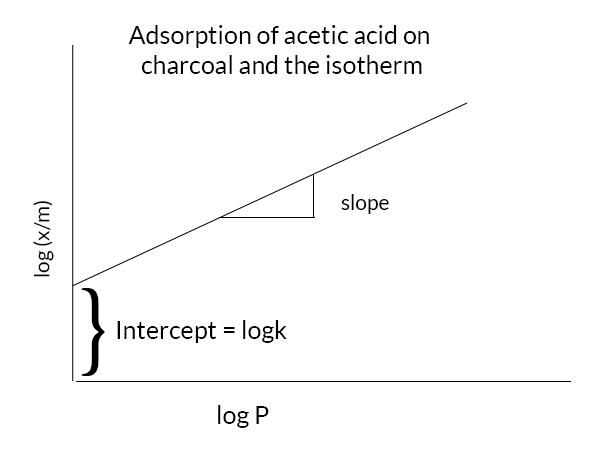
How to decide whether an adsorption in solution follows the Langmuir or Freundlich isotherm? | ResearchGate
Analysis of Selected Methods Use for Calculation of the Coefficients of Adsorption Isotherms and Simplified Equations of Adsorpt

Adsorption Equilibrium and Kinetics of the Removal of Ammoniacal Nitrogen by Zeolite X/Activated Carbon Composite Synthesized from Elutrilithe

4. Adsorption Equilibria Examples - Example 1 An adsorption study is set up in laboratory by adding a known amount of activated carbon to six flasks | Course Hero

Figure 6 from Synthesis of poly(ortho-phenylenediamine) fluffy microspheres and application for the removal of Cr(VI) | Semantic Scholar

Recent trends on density functional theory–assisted calculations of structures and properties of metal–organic frameworks and metal–organic frameworks-derived nanocarbons | Journal of Materials Research | Cambridge Core

SOLVED: Question 12 2 pts Adsorption of Acetic Acid bYActivated Carbonin Aqueous Medium experiment the slope of polt of Celqe versus Ce = 0.0031 and the intercept = 0.0375, Calculate the Adsorption capacity at equilibrium; Qo 0.0031 0.0375 116.7 322.6 911
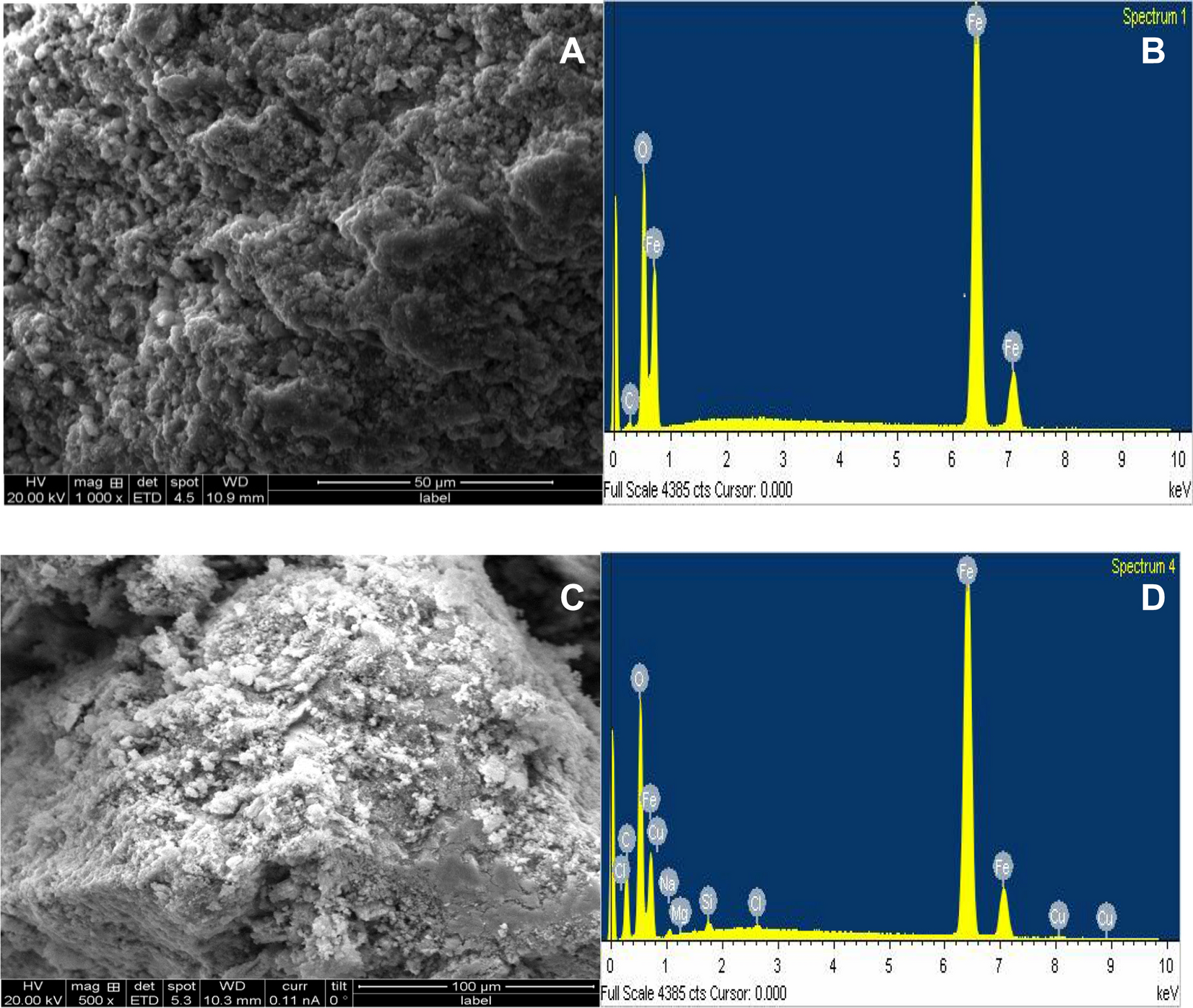
Two–three parameters isotherm modeling, kinetics with statistical validity, desorption and thermodynamic studies of adsorption of Cu(II) ions onto zerovalent iron nanoparticles | Scientific Reports

Comparison of Four Adsorption Isotherm Models for Characterizing Molecular Recognition of Individual Phenolic Compounds in Porous Tailor-Made Molecularly Imprinted Polymer Films | ACS Applied Materials & Interfaces

How to apply Linear Form of Langmuir Isotherma Method on experimental data using Origin Software - YouTube

How to decide whether an adsorption in solution follows the Langmuir or Freundlich isotherm? | ResearchGate
How to Calculate Adsorption Isotherms of Particles Using Two-Parameter Monolayer Adsorption Models and Equations Indonesian Jour
Analysis of Selected Methods Use for Calculation of the Coefficients of Adsorption Isotherms and Simplified Equations of Adsorpt
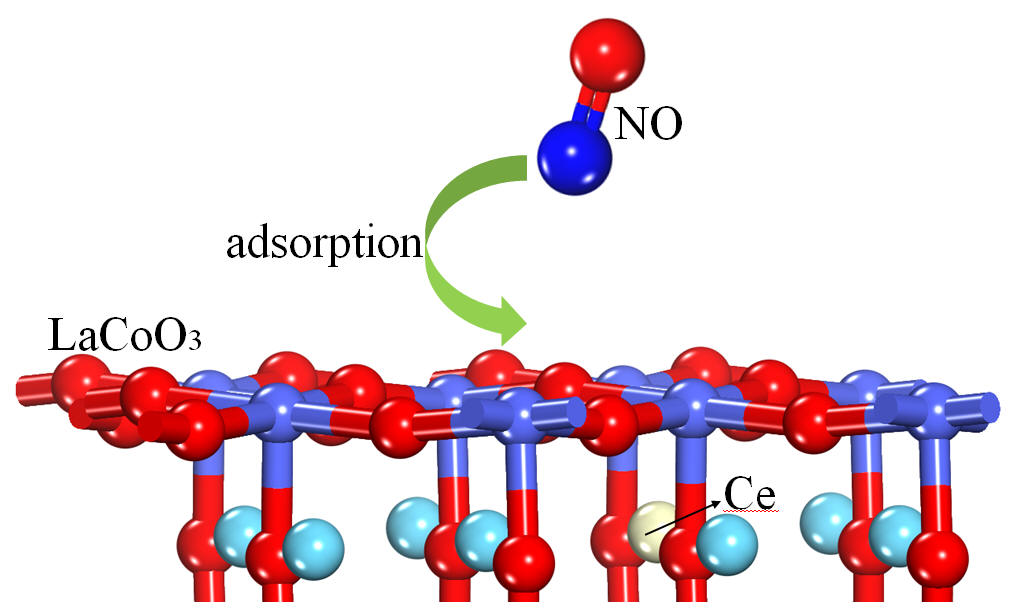
Materials | Free Full-Text | DFT Analysis of NO Adsorption on the Undoped and Ce-Doped LaCoO3 (011) Surface


%20of%20langmuir%20isotherm.jpg?ezimgfmt=rs:363x257/rscb1/ngcb1/notWebP)