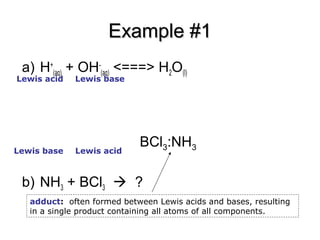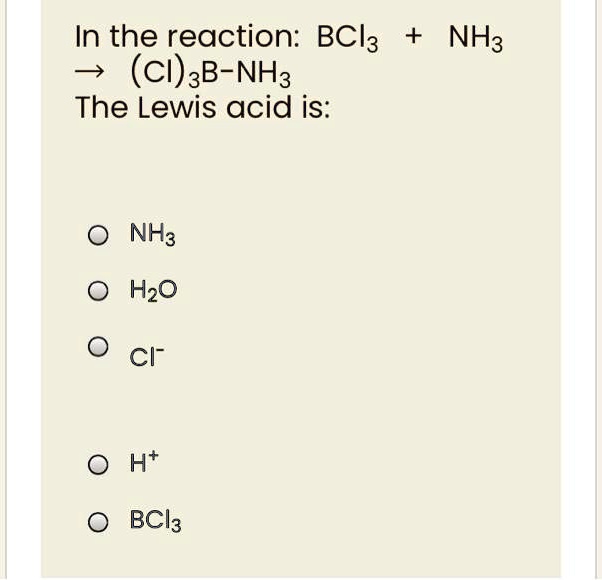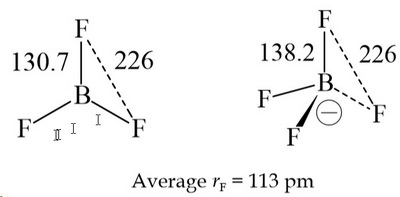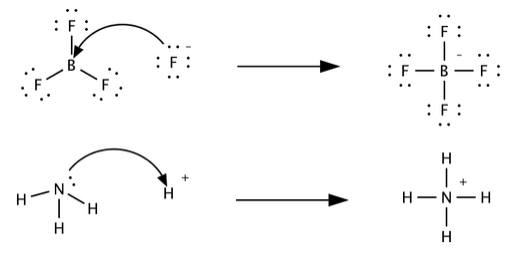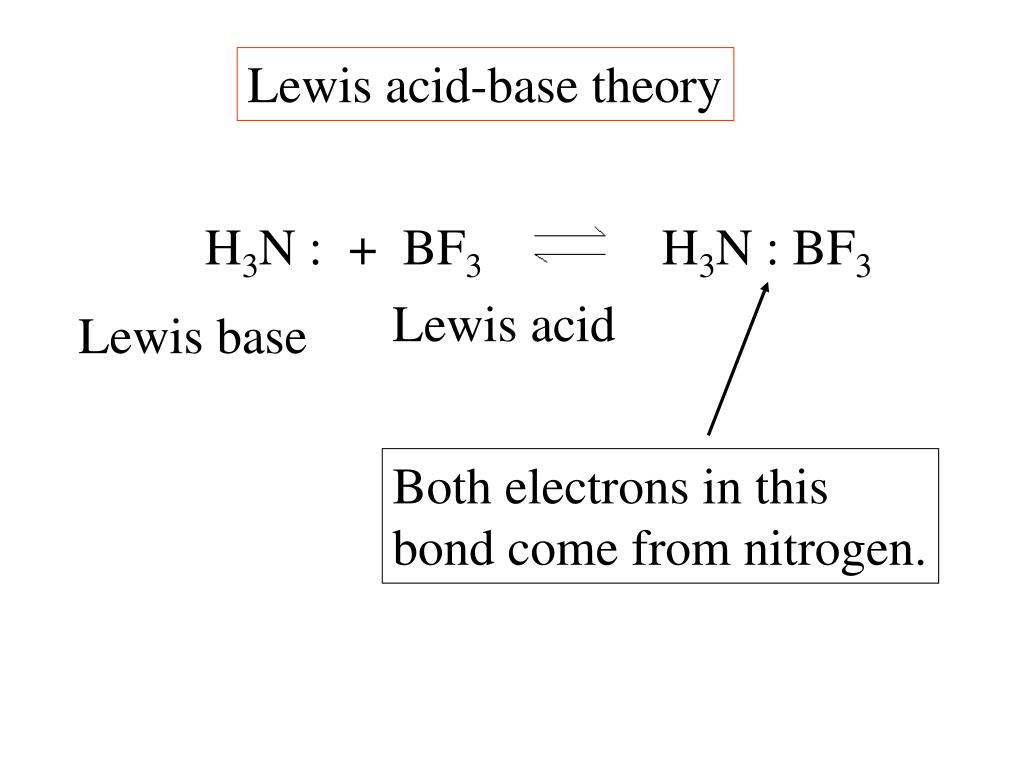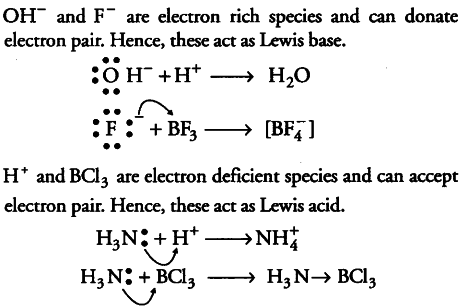
Classify the following species into Lewis acids and Lewis bases and show how these act as Lewis acid/base? - CBSE Class 11 Chemistry - Learn CBSE Forum
![SOLVED: BCl3 + Cl- = [ BCl 4]- , in the equation which is the base? * a. BCl3 b. Cl c. BCl 4 d. none of these. SOLVED: BCl3 + Cl- = [ BCl 4]- , in the equation which is the base? * a. BCl3 b. Cl c. BCl 4 d. none of these.](https://cdn.numerade.com/ask_previews/39f5fa0a-dd33-4254-bfb2-4ac92f81a9c1_large.jpg)
SOLVED: BCl3 + Cl- = [ BCl 4]- , in the equation which is the base? * a. BCl3 b. Cl c. BCl 4 d. none of these.

Classify the following species into Lewis acids and Lewis bases and show how these act as Lewis ... - YouTube