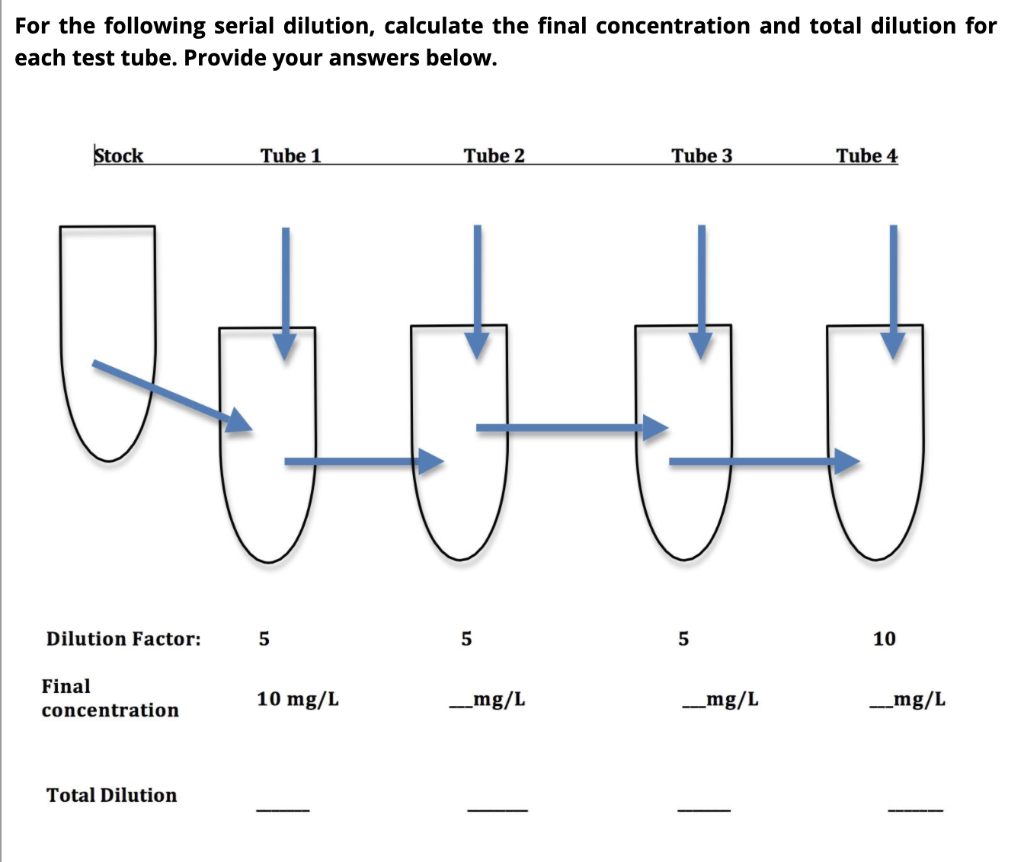
SOLVED: DILUTION AND CONCENTRATION When liquid medication of a given strength is diluted, its strength will be reduced. For example, 10 mL of a solution containing g of a substance has strength

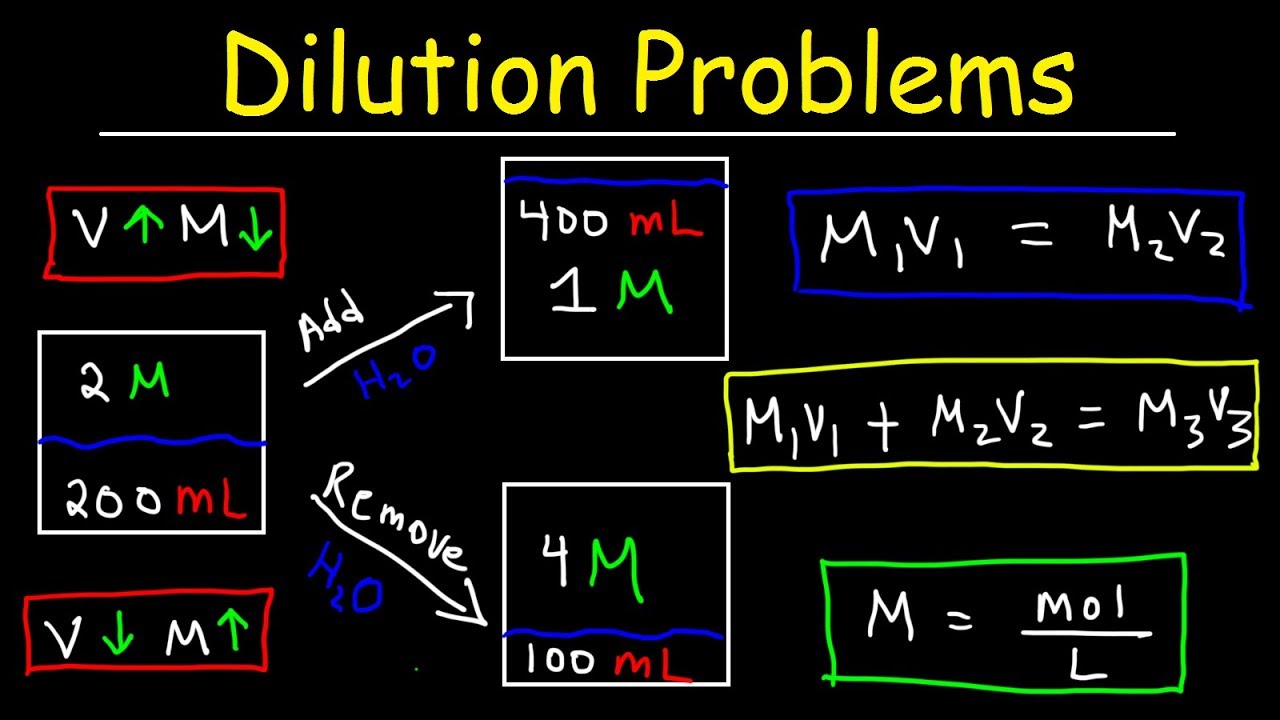
Dilutions. Solve problems involving the dilution of solutions. Include: dilution of stock solutions, mixing common solutions with different volumes and. - ppt download

Dilution Equation & Examples | How to Calculate Dilution Factors - Video & Lesson Transcript | Study.com

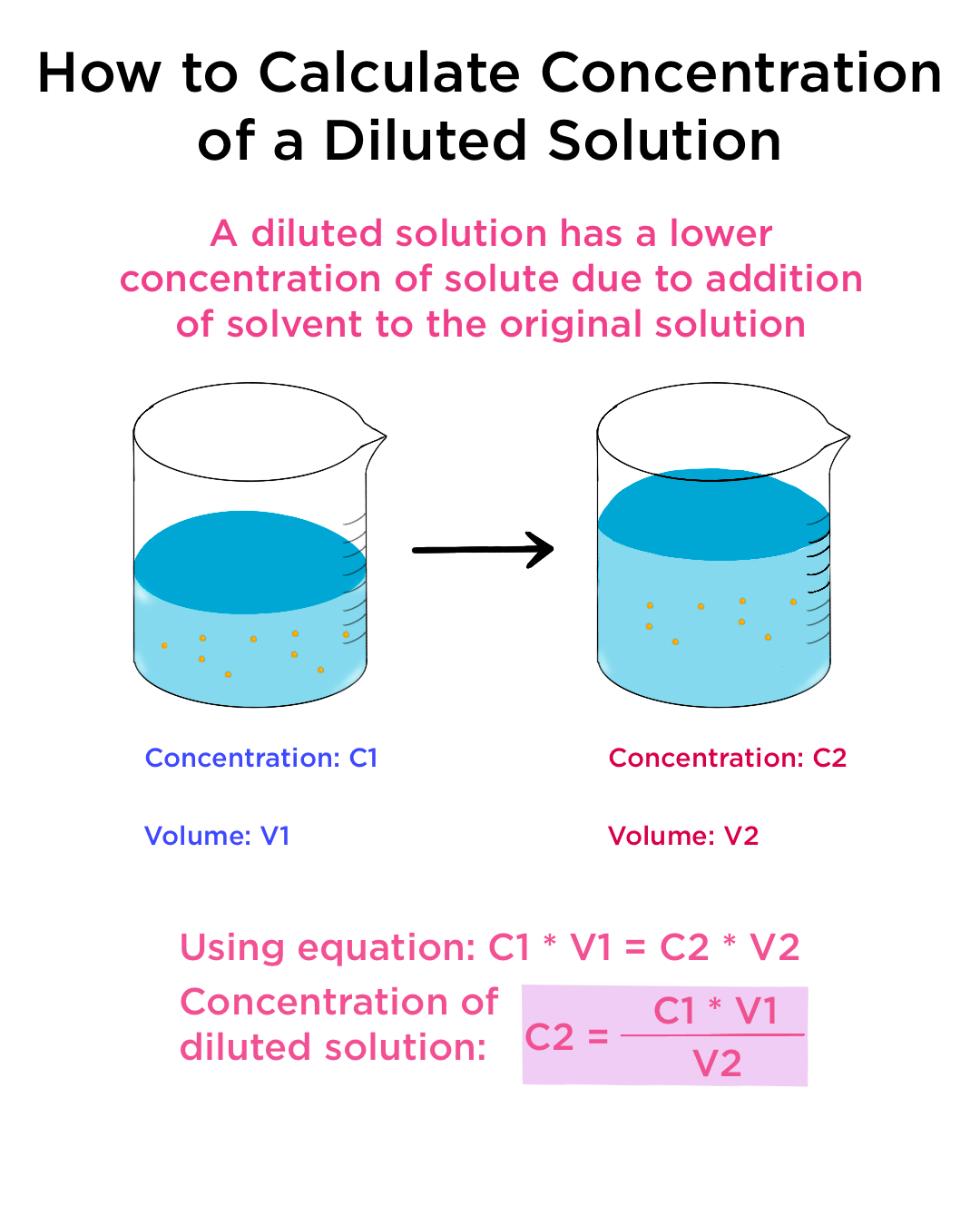
Solution Dilutions. D ilution In a dilution water is added. volume increases. concentration decreases. Copyright © 2009 by Pearson Education, Inc. - ppt download